Background: Monoclonal B-cell lymphocytosis (MBL) is a precursor condition to chronic lymphocytic leukemia (CLL) characterized by the presence of circulating monoclonal B-cell populations in the peripheral blood of otherwise healthy individuals. MBL increases risk of several adverse clinical outcomes, including development of hematological malignancies. Mosaic chromosomal alterations (mCAs) are a form of clonal hematopoiesis which affect large segments of DNA and include gains, losses, and copy number neutral loss of heterozygosity events. mCAs can be detected from SNP-array genotyping. Prior work has suggested mCAs as a potent risk factor for hematological malignancies. However, the nature of the relationship between mCAs and MBL is unknown. We sought to determine 1) the association between mCAs and MBL and 2) their combined effect on risk of hematological malignancies, in a large, prospectively collected, and well-annotated cohort of individuals with known MBL, mCA, and clinical outcomes.
Methods: We examined the prevalence of mCA in a cohort of individuals from the Mayo Clinic Biobank who were screened for MBL using a sensitive eight-color flow cytometry assay. We classified MBL by the size of the clone relative to total B-cells, with high count (HC) defined as those individuals who had a percent clonal B-cell count ≥85% out of total B-cell count and low-count (LC) otherwise. Autosomal mCAs were detected from whole blood DNA collected at the time of MBL screening using a SNP-array-based assay. The MoChA pipeline was used to identify mCA events. Logistic regression was used to estimate odds ratios (OR) and 95% confidence intervals (CIs). To examine incident hematological cancers, we identified and validated all hematological cancers through medical record abstraction using the International Statistical Classification of Diseases codes. Cox regression was used to calculate hazard ratios (HR) and 95% CIs, stratified by MBL status or autosomal mCA status. All models were adjusted for age and sex.
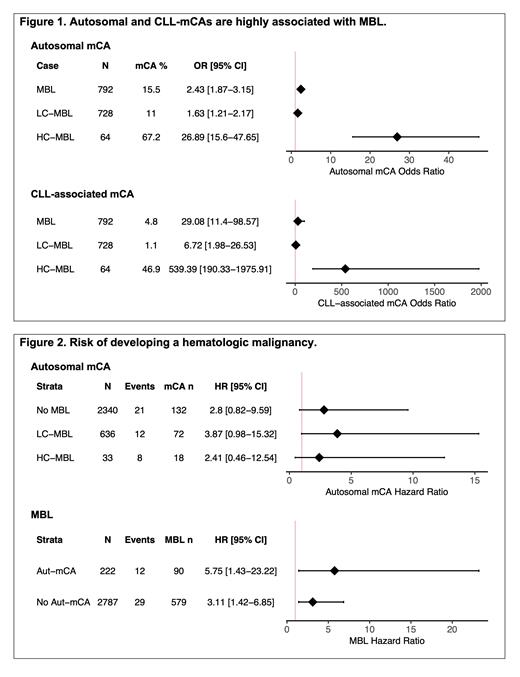
Results: We evaluated the prevalence of autosomal mCA in 3,764 individuals (792 individuals with MBL of whom 64 had HC-MBL and 2,972 without MBL). The prevalence of autosomal mCA was 7.7% ( n=288) with a median mCA cell fraction of 0.09 (range 0.01-1). Among those with and without MBL overall, the prevalence of mCA was 15.5% ( n=123) and 5.6% ( n=165), respectively (Figure 1). Compared to those without an autosomal mCA, individuals with an autosomal mCA were 2.4-fold more likely to have MBL (95% CI 1.9-3.2; P =2.0x10 -11; Figure 1). CLL-associated mCA (i.e., those commonly altered in CLL including trisomy 12 and deletion of 6q, 11q, 13q, and 17p) were present in 42 individuals, of whom 38 (90.5%) had MBL overall and 30 (71.4%) individuals had HC-MBL. Compared to those without a CLL-associated mCA, individuals with a CLL-associated mCA were 539.39-fold more likely to have HC-MBL (95% CI 190.3-1975.9; P <2.0x10 -16) and 6.7-fold more likely to have LC-MBL (95% CI 2.0-26.5; P =0.0031; Figure 1).
Next, we evaluated the relationship between MBL and mCA with risk of developing a hematologic malignancy in 3,009 individuals with follow-up data (median 10 years). Forty-one individuals developed an incident hematological malignancy (68.3% lymphoid). When we evaluated the effect of autosomal mCA stratified by MBL status we observed elevated but non-significant risk of hematological malignancies (Figure 2). When we evaluated the effect of MBL stratified by autosomal mCA status we observed elevated and significant increased risk of developing a hematological malignancy (Figure 2).
Conclusions: We found that the presence of an autosomal mCA is highly associated with having MBL. In particular, >90% of CLL-associated mCA carriers had MBL with ~7- and ~540-fold increased association for LC- and HC-MBL, respectively. These results suggest that almost all individuals in large biobanks with CLL-associated mCAs have undiagnosed MBL. However, not all individuals with MBL have an mCA.
When evaluating risk of hematological malignancies, we found autosomal mCAs had >2-fold elevated but non-significant effects when stratified by MBL status while MBL remained a significant risk factor regardless of autosomal mCA status. Collectively these results support that MBL remains an important risk factor and suggests that mCAs hold potential to identify a subgroup of individuals at greater risk of developing a hematological malignancy.
Disclosures
Sekar:Vertex Pharmaceuticals: Other: Stock . Parikh:AbbVie Inc: Membership on an entity's Board of Directors or advisory committees, Research Funding; AstraZeneca: Membership on an entity's Board of Directors or advisory committees; Agios Pharmaceuticals: Membership on an entity's Board of Directors or advisory committees; Accerta Pharmaceuticals: Research Funding; Bristol Myers Squibb-Celgene: Membership on an entity's Board of Directors or advisory committees, Research Funding; Dava Oncology: Membership on an entity's Board of Directors or advisory committees; Janssen: Membership on an entity's Board of Directors or advisory committees; Boehringer Ingelheim Pharmaceuticals Incc: Membership on an entity's Board of Directors or advisory committees; Vincerx: Research Funding; CSL Behring: Membership on an entity's Board of Directors or advisory committees; Beigene: Membership on an entity's Board of Directors or advisory committees; Juno Therapeutics: Membership on an entity's Board of Directors or advisory committees; Pharmacyclics: Membership on an entity's Board of Directors or advisory committees, Research Funding; Dren Bio: Membership on an entity's Board of Directors or advisory committees; Genentech: Research Funding; Sunesis: Research Funding. Kay:Abbvie: Membership on an entity's Board of Directors or advisory committees, Research Funding; Acerta Pharma: Research Funding; Bristol Meyer Squib / Celgene: Membership on an entity's Board of Directors or advisory committees, Research Funding; Genentech: Research Funding; Pharmcyclics: Membership on an entity's Board of Directors or advisory committees, Research Funding; Sunesis: Research Funding; Vincerx: Research Funding; Agios Pharm: Membership on an entity's Board of Directors or advisory committees; Astra Zeneca: Membership on an entity's Board of Directors or advisory committees; Dren Bio: Membership on an entity's Board of Directors or advisory committees; Janssen: Membership on an entity's Board of Directors or advisory committees; Behring: Membership on an entity's Board of Directors or advisory committees; Beigene: Membership on an entity's Board of Directors or advisory committees; boehringer ingelheim: Membership on an entity's Board of Directors or advisory committees; Dava Oncology: Membership on an entity's Board of Directors or advisory committees; Juno Therapeutics: Membership on an entity's Board of Directors or advisory committees. Cerhan:NanoString: Research Funding; Protagonist: Other: Safety Monitoring Committee; BMS: Membership on an entity's Board of Directors or advisory committees, Research Funding; Genmab: Research Funding; Genentech: Research Funding. Shanafelt:Pharmacyclics: Research Funding; AbbVie: Research Funding; Genentech: Research Funding. Ebert:TenSixteen Bio: Current equity holder in private company, Membership on an entity's Board of Directors or advisory committees; Skyhawk Therapeutics: Current equity holder in private company, Membership on an entity's Board of Directors or advisory committees; Exo Therapeutics: Current equity holder in private company, Membership on an entity's Board of Directors or advisory committees; Abbvie: Consultancy; Neomorph Inc.: Current equity holder in private company, Membership on an entity's Board of Directors or advisory committees; Novartis: Research Funding; Calico: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal